What is a Medical Physicist?
Radformation’s take on what it means to be a medical physicist.
FLASH-RT, a glimpse into the future of high dose rate radiation oncology. What is it, where it is now, and what lies in store?
Of course, studies have indicated that cancers with low α/β ratios respond better to high dose (2+ Gy/Fraction) hypofractionated treatments. Since the delivered dose is critical in tumor sterilization, many contemporary techniques such as VMAT, adaptive radiotherapy, and hypofractionation seek to increase the fractional dose to the target while also striving for a tighter margin. However, these delivery techniques lead to a low dose bath to surrounding healthy tissue volumes. One promising new solution to this phenomenon is FLASH radiotherapy.
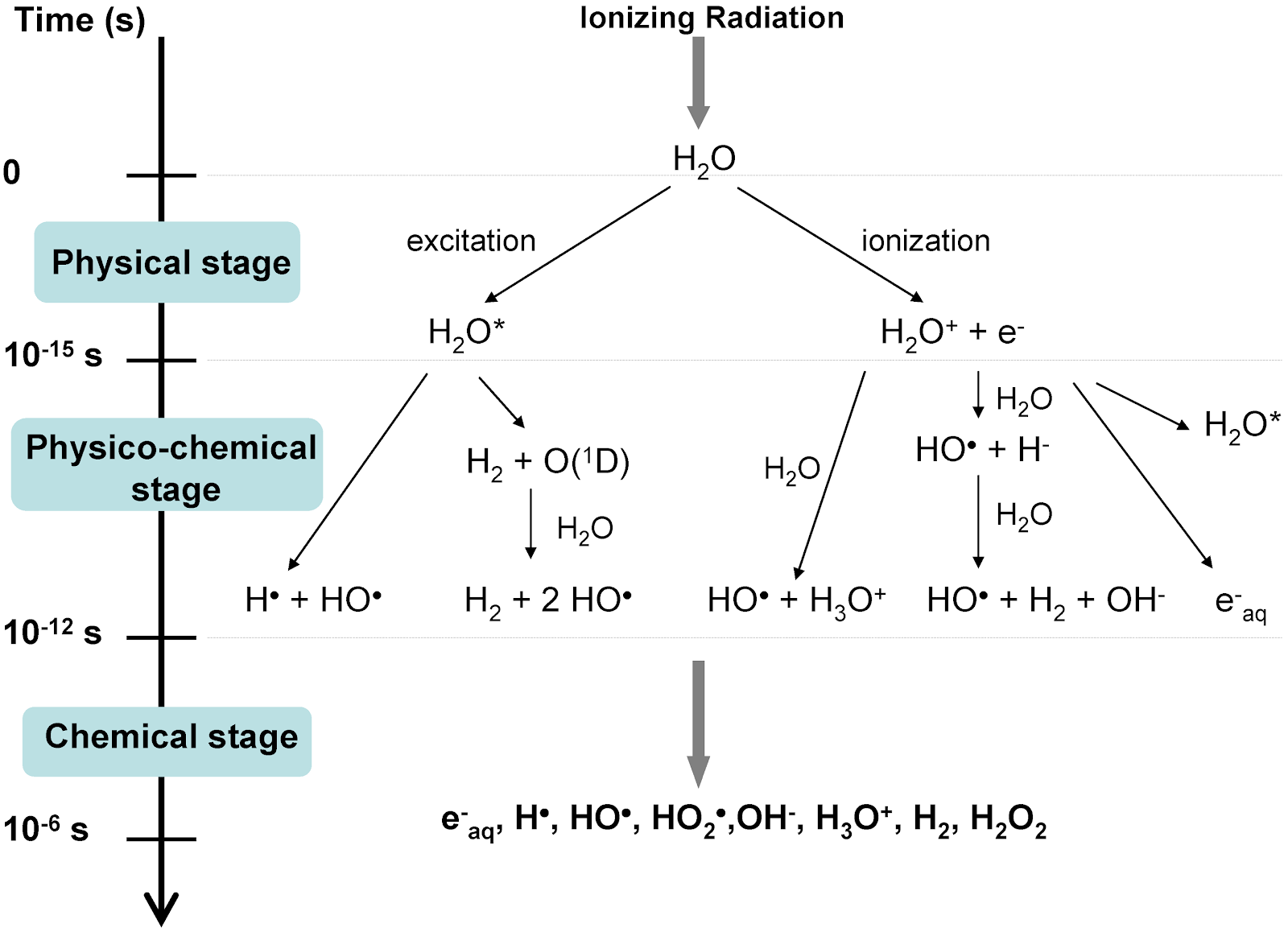
Hornsey and Alper attributed the viability of cells irradiated with ultra-high dose rates to a state of induced transient hypoxia, which minimized the yield of reactive oxygen species (ROS). Hypoxic states protect against low-LET radiation since oxygen acts as a radiosensitizer. ROS pose a problem in healthy tissues as they damage DNA and RNA and can oxidize amino acids and enzymes, leading to a breakdown in cellular function. Unfortunately, their work was left mostly unstudied since many thought the doses needed to induce hypoxia locally were too high.
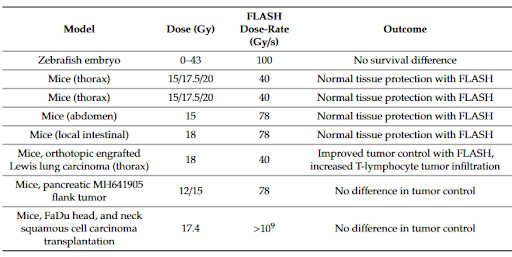
Favaudon et al. reversed this belief in 2014 when they described a similar dose rate effect on physiological response in mice treated using conventional dose rates (0.3 Gy/s) and ultra-high dose rates (40 Gy/s). Their results indicated that FLASH showed similar repression of tumor growth while also significantly reducing the occurrence and severity of early and late-stage acute toxicity. Unlike Hornsey and Alper, Favaudon et al. did not attribute the effects to a state of transient hypoxia. Instead, they suggested a change in DNA repair signaling might play a role.
Their findings sparked a resurgence of research on the subject to unravel the mysterious mechanism(s) behind these effects. To date, despite 3000 papers having been published on the topic, the community does not appear to have arrived at a consensus. According to Dr. Emil Schüler, who co-leads the FLASH initiative at MD Anderson, “While the oxygen hypothesis gets a lot of attention, it cannot be uniquely responsible for the effects we see since oxygenated tumors undergoing FLASH are still responding similarly to conventional delivery. It is likely due to an interplay of oxygen depletion, reactive oxygen species and lymphocyte sparing.”
Another topic of clinical interest is dosimetry. With such high doses, ion chambers saturate and diodes fail, leaving only film, TLDs, and other dose rate independent methods for measurement. To deliver a specific dose, film is used to determine the dose per pulse and the desired number of pulses must be calculated since MU chambers saturate. As we look towards the future and clinical implementation, QA and dosimetry will be important areas of investigation.

Kevin interned with Radformation as a medical physics MS student at Hofstra University, and graduated in May 2021. Kevin’s work mostly focuses on optimizing tasks through programming, since as Church-Turing proposed, “Any effective procedure can be simulated by a (generalized) computer.” His hobbies include cooking, rock climbing (when not trapped on the complanate surface that is Long Island), and video games.
Related tags: RadOnc News
Radformation’s take on what it means to be a medical physicist.
Check out Radformation’s favorite podcasts in radiation therapy.
Radformation’s medical physicists break down highlights from the 2018 ASTRO Guidelines for Whole Breast and discuss how EZFluence automation allows...
Leave a comment